In May 2025, Aktiia closed an oversubscribed $42 million Series B funding round co-led by Earlybird Health and Wellington Partners, bringing total financing to date above $100 million, and simultaneously rebranded its product portfolio to Hilo. In July 2025, the company secured the first-ever U.S. FDA 510(k) clearance for an over-the-counter (OTC) cuffless blood pressure monitor—the G0 Blood Pressure Monitoring System (Hilo Band)—marking a global regulatory first. In November 2024, Hilo received Health Canada approval, its first expansion outside Europe. In 2025, the company also entered Australia and Saudi Arabia, published its inaugural UK Blood Pressure Report: A Nation Under Pressure, and became a Gold Prevention Partner of the British & Irish Hypertension Society (BIHS). The Hilo Band is expected to debut in the U.S. market in 2026.
Company Profile (Updated April 2026)
Aktiia S.A.(Aktiia公司) is a Swiss digital health and medical device company dedicated to revolutionizing hypertension management through continuous, cuffless blood pressure monitoring technology. Headquartered in Neuchâtel, Switzerland, the company was founded in May 2018 by Dr. Josep Solà, Dr. Mattia Bertschi, and Raghav "Rags" Gupta as a spin-out from the Swiss Centre for Electronics and Microtechnology (CSEM), building on nearly two decades of Foundational research in optical biosensing. Aktiia has established itself as the global leader in the emerging cuffless blood pressure monitoring (CBPM) category, with its products trusted by over 130,000 users across 12 markets worldwide.
Development History:
- 2004–2018: Founders Dr. Josep Solà and Dr. Mattia Bertschi spent 15 years at CSEM specializing in decoding heart signals and developing non-invasive blood pressure monitoring technology, laying the scientific foundation for Aktiia
- 2018: Aktiia S.A. founded in Neuchâtel, Switzerland in May as a CSEM spin-out, with the mission to tackle the global hypertension crisis through wearAble technology
- 2018: Raised CHF 4 million Seed round led by Redalpine and TransLink Capital
- 2020: Raised CHF 6.1 million ($6.1M) to bring its cuffless optical blood pressure monitor to market
- 2021: Raised $17.5 million Series A led by Draper Esprit, with participation from Redalpine, 415 Capital, Verve Ventures, and Translink Capital; CE Mark approved as Class IIa medical device for adult blood pressure monitoring in Europe; launched first 24/7 blood pressure monitoring device in the UK
- 2024: Completed CHF 27 million ($30 million) funding round led by Redalpine, with Khosla Ventures, Molten Ventures, Translink Capital, and Verve Ventures; received CE Mark for CALFREE calibration-free technology, enabling blood pressure data collection via optical sensors in smartwatches and smartphone cameras without cuff calibration; secured Health Canada approval in November
- 2025: In May, closed $42 million Series B (oversubscribed) co-led by Earlybird Health and Wellington Partners, with new investors Kfund and naturalX Health Ventures, bringing total funding above $100 million; rebranded product portfolio to Hilo; ACHieved 76% annual revenue growth; in July, secured FDA 510(k) clearance for OTC cuffless BP monitor (global first); entered Australia and Saudi Arabia; published UK Blood Pressure Report: A Nation Under Pressure; became Gold Prevention Partner of BIHS
- 2026: Preparing for U.S. market launch of Hilo Band; continuing to expand regulatory pathways and market access; advancing integration of Hilo into clinical care pathways and enterprise use cases
Core Mission: To manage the world's blood pressure through innovation, accessibility, and scientific rigor—making healthy blood pressure achievable for everyone by transforming hypertension from an intermittent, inconvenient measurement into a continuous, effortless, and actionable health insight.
Technology Platform & Product Portfolio
Hilo Band (G0 Blood Pressure Monitoring System)
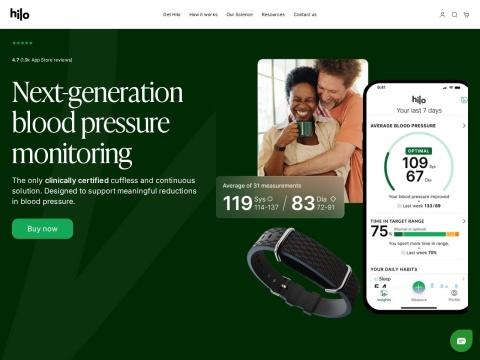
The Hilo Band is Aktiia's flagship wrist-worn, cuffless blood pressure monitor. It utilizes photoplethysmography (PPG) optical sensors to collect signals from arteries in the wrist, combined with proprietary Pulse wave analysis (PWA) algorithms to estimate blood pressure. The device measures the unique pressure pattern generated by each heartbeat as it moves through the wearer's blood vessels.
Key Features:
- Cuffless & Continuous: Automatically takes blood pressure reADINgs throughout the day and night, but only when the user is at rest, ensuring accuracy without lifestyle disruption
- Initial Calibration: Users calibrate the device once using a provided traditional cuff; thereafter, no cuff is required for ongoing measurements
- Form Factor: Sleek, lightweight, bracelet-style wearable designed for comfort and everyday use, available with interchangeable sport and leather bands
- Hilo Companion App: Integrated smartphone app that displays blood pressure trends, patterns, and insights; data is sent to the app and cloud server for algorithmic analysis
- Battery Life: Up to 15 days of continuous use; full charge takes approximately 90 minutes
- Reading Frequency: Provides over 800 readings per month, compared to approximately 1–2 readings per month with traditional cuff-based home monitoring
- Clinical Validation: Supported by a pivotal clinical trial comparing the G0 system against double auscultation (gold standard) in 140 patients; 91% of hypertension patients in a Massachusetts General Hospital collaboration preferred Hilo over traditional monitoring
- Regulatory Status: CE Marked (Class IIa) since 2021; FDA 510(k) cleared for OTC use (July 2025); Health Canada approved (November 2024); approved in Australia and Saudi Arabia
- Pricing (Europe): £209.99 / €229.99 (approximately $280 USD)
CALFREE Technology
CALFREE is Aktiia's calibration-free optical blood pressure monitoring technology, which received CE Mark approval in Europe in 2024. It allows blood pressure data to be collected using inputs from optical sensors commonly found in consumer devices such as smartwatches and smartphone cameras, without requiring initial cuff calibration. This technology opens the door for third-party integration into wearable devices, cameras, and smart bands.
Blood Pressure Intelligence Platform
Aktiia has developed a proprietary foundation Machine Learning model specifically designed for blood pressure. The model has been trained on tens of billions of optical signals from real-world users and refined with hundreds of millions of calibration points, establishing leadership in the CBPM category. The platform supports both consumer (D2C) and enterprise use cases, including healthcare provider integrations and population health initiatives.
Clinical Evidence & Scientific Foundation
Published Research & Validation
- The company's multidisciplinary team has contributed to over 120 peer-reviewed publications and holds more than 35 patents in optical biosensing and blood pressure estimation
- Clinical trials demonstrate that the Hilo Band's accuracy meets international standards (e.g., ISO 81060-2) for cuffless blood pressure devices
- Real-world data from 130,000+ global users validates the technology's performance across diverse populations and use cases
- In collaboration with Massachusetts General Hospital, 91% of hypertensive patients preferred the Hilo Band's "perception-free" wearability and continuous data over traditional cuff-based monitoring
Financial Performance & Funding History
Funding Rounds (Total >$100 Million)
- Seed (September 2018): CHF 4 million — led by Redalpine and TransLink Capital
- Pre-Series A (March 2020): CHF 6.1 million ($6.1M)
- Series A (November 2021): $17.5 million — led by Draper Esprit, with Redalpine, 415 Capital, Verve Ventures, and Translink Capital
- Series A Extension (February 2024): CHF 27 million ($30 million) — led by Redalpine, with Khosla Ventures, Molten Ventures, Translink Capital, and Verve Ventures
- Series B (May 2025): $42 million (oversubscribed) — co-led by Earlybird Health and Wellington Partners, with new investors Kfund and naturalX Health Ventures, and participation from existing investors redalpine, Khosla Ventures, Molten Ventures, Translink Capital, and Verve Ventures
- Total Financing to Date: Above $100 million
Commercial Metrics (2025)
- Devices Sold: Over 130,000 units globally
- Annual Revenue Growth (2025): 76%
- Global Markets: 12 countries across Europe, North America, Asia-Pacific, and Middle East
- User Retention: Exceeds typical wearable device retention rates
Company Overview
Corporate Information
Company Name: Aktiia S.A. (Legal Name: AKTIIA SA)
Product Brand: Hilo (rebranded from Aktiia in May 2025)
Founded: May 2018
Headquarters: Neuchâtel, Switzerland
Company Type: Private Venture-Backed Company
Industry: Digital Health / Medical Devices / Wearable Technology
Employees: Not publicly disclosed
Leadership Team (2025–2026)
Chief Executive Officer: Raghav "Rags" Gupta
- Co-founder; leads the company's global commercial strategy and mission to scale cuffless blood pressure monitoring worldwide
Co-Founder & Chief Technology Officer: Dr. Josep Solà
- Leads technology development and algorithm innovation; 15+ years of research experience in optical biosensing and cardiovascular signal processing at CSEM; co-architect of the Hilo Band's PPG and pulse wave analysis technology
Co-Founder: Dr. Mattia Bertschi
- Background in biomedical engineering; previously served as CEO during early company stages; continues as co-founder and key leader in scientific and strategic development
Board Director: Dr. Christoph Massner (Earlybird Health Principal)
Strategic Partnerships & Collaborations
Academic & Clinical Partners
- Massachusetts General Hospital (USA): Clinical collaboration demonstrating 91% patient preference for Hilo Band over traditional cuff monitoring
- Swiss Centre for Electronics and Microtechnology (CSEM): Origin of foundational optical biosensing research; ongoing technology development relationship
Professional Society Partnerships
- British & Irish Hypertension Society (BIHS): Gold Prevention Partner (2025), contributing to prevention, evidence-based innovation, and population health initiatives
Enterprise & Distribution
- Building out blood pressure intelligence platform for enterprise use cases, including healthcare systems, insurers, and employer wellness programs
- Third-Party Integration: CALFREE technology enables integration into consumer electronics (smartwatches, smartphone cameras, smart bands) through licensing partnerships
Global Market & Competitive Landscape
Target Markets
Europe: Core market with CE Mark approval since 2021; strong presence in the UK, Germany, Switzerland, and EU member states; published UK Blood Pressure Report to support national hypertension prevention efforts
North America: Priority expansion market; FDA OTC clearance secured July 2025; Health Canada approval obtained November 2024; U.S. commercial launch planned for 2026
Asia-Pacific: Launched in Australia in 2025; exploring additional markets in the region
Middle East: Approved and launched in Saudi Arabia in 2025
Primary Competitors
- Samsung Galaxy Watch (South Korea): FDA-authorized for blood pressure trend monitoring with periodic cuff calibration; not cleared for clinical diagnosis
- Withings ScanWatch (France): CE-marked hybrid analog-digital watch with PPG-based cardiovascular monitoring
- Valencell (USA): Cuffless and calibration-free fingertip blood pressure monitor (January 2024 unveiling)
- Omron (Japan): Traditional cuff-based blood pressure monitors with wearable extensions
- Apple Watch (USA): Does not include blood pressure monitoring as of 2026, despite market speculation
- BioBeat (Israel): G-7 Continuous Patch adhesive wearable for multi-vital monitoring
Competitive Advantages
- First-Mover Regulatory Position: First and only FDA-cleared OTC cuffless blood pressure monitor globally; CE Marked since 2021 with extensive real-world validation
- Proprietary ML Foundation Model: Trained on tens of billions of optical signals and hundreds of millions of calibration points from 130,000+ users—one of the largest cuffless BP datasets in existence
- Dual-Mode Technology: Both cuffless wrist-worn device (Hilo Band) and calibration-free sensor technology (CALFREE) for third-party integration
- Scientific Pedigree: 20 years of CSEM research heritage; 120+ peer-reviewed publications; 35+ patents
- Consumer-Centric Design: Medical-grade accuracy in a sleek, bracelet-style form factor with high user preference (91% vs. traditional cuffs) and retention rates exceeding typical wearables
- Enterprise SCalability: Blood Pressure Intelligence Platform supports both direct-to-consumer and B2B/healthcare system deployments
2026 Outlook & Strategic Priorities
Near-Term Objectives
- U.S. Market Launch: Execute commercial launch of Hilo Band in the United States following FDA OTC clearance, initially via online direct-to-consumer channels
- Clinical Care Pathway Integration: Integrate Hilo into hypertension management protocols, remote patient monitoring programs, and value-based care models
- Enterprise Platform Expansion: Scale blood pressure intelligence platform for healthcare providers, payers, and employer wellness programs
- Third-Party CALFREE Licensing: Partner with smartwatch, smartphone, and wearable manufacturers to integrate calibration-free blood pressure technology into consumer electronics
- Regulatory Expansion: Pursue additional market approvals in Asia-Pacific, Latin America, and Middle East
- Prevention Research: Advance long-term cardiovascular prevention research using real-world continuous blood pressure data
Contact Information
Corporate Headquarters
Address: Neuchâtel, Switzerland
Online Presence
Corporate Website: www.hilo.com
Former Brand Website: www.aktiia.com
Keywords: Aktiia,Hilo,Hilo Band,G0 Blood Pressure Monitoring System,cuffless blood pressure monitor,continuous blood pressure monitoring,PPG,photoplethysmography,pulse wave analysis,OBPM,optical blood pressure monitoring,hypertension,wearable medical device,CE Mark,FDA 510(k),OTC,Health Canada,Neuchâtel,Switzerland,CSEM,Josep Sola,Mattia Bertschi,Raghav Gupta,Earlybird Health,Wellington Partners,Series B,CALFREE,blood pressure intelligence,BIHS,UK Blood Pressure Report,A Nation Under Pressure