
📢 Key Milestones (2024–2026):
In August 2024, NanoHive Medical raised $7 million in Series C financing to accelerate commercial growth and innovation. In June 2025, the company secured an exclusive sublicense of DirectSync Surgical's piezoelectric implantAble sensor technology for spinal fusion, exploring the development of smart spinal implants. In April 2026, NanoHive announced an exclusive strategic partnership with Canary Medical to jointly develop and commercialize next-generation smart spinal cages that integrate Canary's implantable sensing technology with NanoHive's Hive™ Soft Titanium® 3D printed spinal implant design.
Company Profile (Updated April 2026)
NanoHive Medical, LLC is a commercial-stage medical technology company headquartered in Woburn, Massachusetts, USA. Originally founded in 2014 as HD LifeSciences LLC, the company rebranded to NanoHive Medical and has established itself as a pioneer and leADINg innovator in 3D printed spinal interbody fusion implants and instrumentation.
The company's mission is to be the leader in spinal fusion personalization by leveraging its proprietary Hive™ Soft Titanium® interbody fusion lattice technology. NanoHive's biomimetic 3D printed titanium implants are designed to provide surgeons and patients with ideal biomechanical elastic modulus properties comparable to PEEK, clear and precise diagnostic imaging capability, and optimal osteoblast cell attraction and integration — all features that lead to consistently strong fusion constructs and efficacious clinical experiences.
Development History:
- 2014: Founded as HD LifeSciences LLC in Woburn, MassACHusetts
- 2021: Rebranded to NanoHive Medical, LLC; launched Hive Standalone ALIF System, a fixated Soft Titanium latticed 3D printed titanium implant system for anterior lumbar interbody fusion
- 2022: Expanded patent portfolio with issuance of US Patent 11,253,368 B2 for methods of designing high X-ray lucency lattice structures; 16th issued patent for the company
- 2023: Received FDA 510(k) clearance for 22mm length Hive™ PL Interbody System, expanding the posterior lumbar interbody fusion portfolio
- 2023.12: Closed a venture debt round provided by Kenston Capital Partners, LLC, with Outcome Capital, LLC acting as strategic and financial advisor
- 2024.04: Entered into a distribution agreement with HC Trade USA to expand into new international markets
- 2024.08: Completed $7 million Series C financing to capitalize Rapid growth and prioritize company-building to profitability; welcomed Greg Hoffman and David Scott to the Board of Directors
- 2025.06: Secured exclusive sublicense of DirectSync Surgical's piezoelectric implantable sensor technology (licensed from the University of Kansas) for the field of spinal fusion; exploring development of bone-stimulating and remote monitoring/data collection sensor technology housed in the Hive Soft Titanium spinal interbody device lattice
- 2026.04: Announced exclusive strategic partnership with Canary Medical to jointly develop next-generation smart spinal cages integrating Canary's proprietary implantable sensing technology with NanoHive's Hive™ Soft Titanium® spinal implant design capabilities
Core Mission: To be the leader in spinal fusion personalization by leveraging innovative 3D printed titanium lattice technology to deliver best-in-class spine interbody devices that enhance patient healing and treatment efficacy.
Core Product Portfolio
Hive™ Standalone ALIF System
- Approach: Anterior Lumbar Interbody Fusion (ALIF)
- Design: Fixated, Soft Titanium latticed 3D printed titanium implant system indicated for one and two-level ALIF procedures
- Features: Available in various footprints and lordotic angles; aggressive screws and endplates for fixation; unique graft channels allowing injection of additional bone graft into lateral walls and central lumen
Hive™ PL Interbody System (TLIF/PLIF)
- Approach: Posterior Lumbar Interbody Fusion (PLIF/TLIF)
- Design: Latticed titanium interbody devices for posterior lumbar approaches in multiple sizes and geometries
- Expansion: 22mm length line extension cleared by FDA in 2023
Hive™ Cervical System
- Approach: Cervical Interbody Fusion
- Options: Zero-profile standalone devices with self-tapping screw fixation, and cage-plus-plate configurations
- Variety: Multiple footprints, heights, and lordoses available
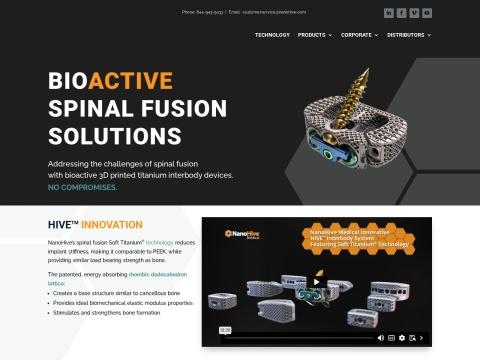
Proprietary Technology: Soft Titanium®
- Lattice Structure: Patented rhombic dodecahedron cell structure creating a base structure similar to cancellous bone
- Biomechanics: Ideal elastic modulus properties comparable to PEEK, reducing stress shielding
- Imaging: Enhanced radiographic visibility (X-ray lucency) allowing physicians to evaluate bone growth through the titanium implant without traditional lateral viewing windows
- Biology: Optimized porosity and surface architecture for osteoblast cell attraction, ingrowth, and ongrowth
- Manufacturing: Additively manufactured via partnership with 3D Systems, leveraging over 2 million medical devices printed and FDA-registered, ISO-certified facilities
Strategic Partnerships
Canary Medical (Smart Spinal Cages — 2026)
- Exclusive Partnership: Joint development of next-generation smart spinal cages integrating Canary Medical's proprietary implantable sensing technology with NanoHive's Hive™ Soft Titanium® spinal implant design capabilities
- Objective: Deliver objective, actionable data on fusion progression, segment mobility, and patient activity through embedded sensing technology in spinal interbody fusion cages
- Market: Global spinal fusion market estimated at $6–$9 billion today, projected to exceed $13–$16 billion over the next decade
DirectSync Surgical (Smart Sensor Technology — 2025)
- Technology: Exclusive sublicense of DirectSync's piezoelectric implantable sensor technology (originally licensed from the University of Kansas) for spinal fusion
- Application: Exploring bone-stimulating and remote monitoring/data collection sensor technology housed within the Hive Soft Titanium lattice
- Regulatory Pathway: Confirmed with FDA that the technology will go through the De Novo pathway
3D Systems (Manufacturing Partner)
- Role: Additive manufacturing partner for NanoHive's intricately designed titanium implants
- Capabilities: Over 2 million medical devices printed; two FDA-registered and ISO-certified facilities; over 100 FDA-cleared and CE-marked devices supported
HC Trade USA (Distribution — 2024)
- Agreement: Distribution partnership to represent NanoHive's Hive portfolio in new international markets
Regulatory & Intellectual Property
- FDA 510(k) Clearances: Multiple clearances including Hive Standalone ALIF, Hive PL Interbody System (including 22mm length extension), and Hive Cervical systems
- Patent Portfolio: 16+ issued patents including US 11,253,368 B2 for high X-ray lucency lattice structures
- Smart Implant Regulatory: Piezoelectric sensor technology confirmed for FDA De Novo pathway
Company Overview
Corporate Information
Legal Name: NanoHive Medical, LLC (fka HD LifeSciences LLC)
Founded: 2014
Headquarters: 12 Gill Street, Suite 4500, Woburn, MA 01801, USA
Phone: 844-943-5433
Company Type: Privately held LLC
Industry: Medical Devices / Spinal Orthopedics / 3D Printed Implants
Estimated Employees: 1–50 (small enterprise per industry classification)
Website: www.nanohive.com
Leadership Team
Chief Executive Officer & President: Patrick O'Donnell
- Leads corporate strategy, commercial operations, and investor relations
Co-Founder & Vice President of Research & Development / Chief Technology Officer: Ian Helmar
- Drives implant design innovation, lattice architecture development, and intellectual property expansion
Board of Directors:
- Greg Hoffman: Managing Partner of Hoffco Holdings (appointed August 2024)
- David Scott: Co-founder and former CEO of Xator Corporation (appointed August 2024)
- Sami El-Saden: Existing board member
- Ian Helmar: Co-Founder and VP R&D
- Patrick O'Donnell: CEO & President
Competitive Landscape
- Johnson & Johnson / DePuy Synthes: Global orthopedic and spine market leader
- Medtronic: Comprehensive spinal implant and navigation portfolio
- Stryker: Major competitor in spinal interbody fusion and 3D printed titanium implants (Tritanium)
- Zimmer Biomet: Strategic partner of Canary Medical; competitor in spinal fusion
- Globus Medical: Innovative spine implant and robotics company
- NuVasive: Minimally invasive spine surgery solutions
- Intelligent Implants GmbH: SmartFuse electrotherapeutic spinal fusion system
- Exactech / Statera Medical: Smart reverse shoulder implant development
2026 Strategic Outlook
- Smart Spinal Cage Development: Joint development with Canary Medical focusing on design integration, engineering validation, and defining key clinical applications for data-enabled spinal interbody fusion cages
- Smart Implant Regulatory Advancement: Pursue FDA De Novo pathway for piezoelectric sensor-integrated Hive Soft Titanium implants
- Commercial Expansion: Continue scaling U.S. commercial footprint and expand international distribution through HC Trade USA and other partners
- Product Portfolio Growth: Expand Hive portfolio indications and explore patient-specific implant capabilities
- Profitability Focus: Leverage Series C capital to drive company-building toward sustainable profitability
Contact Information
Global Headquarters
Address: 12 Gill Street, Suite 4500, Woburn, MA 01801, USA
Phone: 844-943-5433
Website: www.nanohive.com
Email: Patrick.odonnell@nanohive.com
Investor & Media Contact
Contact: Patrick O'Donnell, CEO & President
Email: Patrick.odonnell@nanohive.com
Keywords: NanoHive Medical, HD LifeSciences, 3D printed spinal implants, spinal interbody fusion, Soft Titanium, Hive ALIF, Hive PLIF, Hive TLIF, Hive Cervical, Hive PL, smart spinal cage, Canary Medical, DirectSync Surgical, rhombic dodecahedron, lattice structure, biomimetic, osseointegration, radiolucency, X-ray lucency, Woburn, Massachusetts, Patrick O'Donnell, Ian Helmar, 3D Systems, Kenston Capital, Outcome Capital, HC Trade USA, Series C, venture debt, FDA 510(k), De Novo, piezoelectric sensor
You May Also Like
-
NanoHive Medical, LLC - Pioneer in 3D Printed Spinal Interbody Fusion Implants
NanoHive Medical, LLC is a commercial-stage medical technolo... -
Canary Medical Inc. - Pioneer of Implantable Smart Sensor Technology for Healthcare
Canary Medical Inc. is a medical technology company headquar... -
ClearPoint Neuro, Inc. - Global Leader in MRI-Guided Neurosurgery and CNS Drug Delivery
ClearPoint Neuro, Inc. is a global medical device company he... -
BioSino Bio-Technology And Science Inc.(中生北控生物科技股份有限公司) - Leading Chinese IVD Reagent Manufacturer
BioSino Bio-Technology And Science Inc. is a China-based in-... -
Switchback Medical, LLC - Specialized CDMO for Minimally Invasive Medical Devices
Switchback Medical, LLC is a specialized contract developmen... -
BVI Medical - Global Leader in Ophthalmic Surgical Devices
BVI Medical is a diversified global ophthalmic device compan... -
ScreenPoint Medical B.V. - Global Leader in AI-Powered Breast Cancer Detection
ScreenPoint Medical B.V. is a Netherlands-based artificial i... -
ARKRAY Factory Pinghu, Inc.(爱科来医疗科技(平湖)有限公司) - ARKRAY Group China Manufacturing Base
ARKRAY Factory Pinghu, Inc. is the wholly-owned Chines... -
ARKRAY Marketing Shanghai, Inc.(爱科来国际贸易(上海)有限公司) - ARKRAY Group China Sales & Service Headquarters
ARKRAY Marketing Shanghai, Inc. is the wholly-owned Chinese... -
ARKRAY, Inc.(株式会社アークレイ) - Global Leader in In-Vitro Diagnostics and Diabetes Care
ARKRAY, Inc. is a privately held Japanese medical device and... -
ARKRAY Factory, Inc.(株式会社アークレイ ファクトリー) - Global IVD Manufacturing Hub of ARKRAY Group
ARKRAY Factory, Inc. is the flagship manufacturing subsidiar... -
QIAGEN Sciences LLC (fka QIAGEN, Inc.) - U.S. Manufacturing & Operations Hub for Sample to Insight S
QIAGEN Sciences LLC is the primary U.S. operating successor...
网友评论
- Popular Tags
-
- USA
- California
- Germany
- China
- Korea
- Abbott
- Massachusetts
- Shanghai
- Israel
- Illinois
- Japan
- Florida
- ARKRAY
- QIAGEN
- France
- Zhejiang
- British Columbia
- Arizona
- 3M
- Netherlands
- Minnesota
- North Rhine-Westphalia
- Canada
- Michigan
- Ireland
- Tokyo
- Beijing
- Munich
- New York
- Missouri
- Seoul
- Guangdong
- Fujirebio
- Ningbo
- Medtronic
- 爱科来
- Singapore
- Agilent
- Mentor
- Johnson
- Gyeonggi-do
- SynCardia
- NanoHive
- Foresight
- Colorado
- Nyxoah
- Belgium
- Mont-Saint-Guibert
- MicroPort
- Zhimai
- Follow Us
-

Scan the QR code to follow our WeChat Official Account

