
InVera Medical achieved CE Mark approval on February 13, 2026 (notified body: BSI NB 2797 under MDR 2017/745) and FDA 510(k) clearance on March 12, 2026 (announced April 7, K250794) for the InVera Infusion Device. These dual approvals enable limited market release in select US and European centers. 12-month pilot clinical study results will be presented by Dr. Lowell Kabnick at the Charing Cross Vascular Symposium in London on April 21, 2026.
Company Overview (Updated April 2026)
InVera Medical is an Irish medical technology company headquartered in Galway, Ireland. Founded on July 24, 2018 by Stephen Cox (CEO and Co-Founder) and Nigel Phelan (Chief Medical Officer and Co-Founder), the company focuses on advancing minimally invasive technologies for the treatment of chronic venous disease (CVD). As a portfolio company of ATU iHubs (Atlantic Technological University innovation ecosystem), InVera Medical represents the second client company in the hub to achieve FDA clearance.
Mission: To create catheter-based solutions for vein disease with a patient-focused and scientifically grounded approach to medical device innovation, addressing the significant gap in care for the over 120 million people affected by CVD across the US and Europe.
History & Development
Key Milestones:
- July 24, 2018: Company founded in Galway, Ireland (Company Number: 630839)
- 2018-2025: Research and development phase; prototype development of helical coil catheter technology; preclinical and pilot clinical studies
- November 25, 2025: Granted US Patent 12,478,399 for "Method of treating a blood vessel in a subject," invented by Sean Cummins, Stephen Cox, and Nigel Phelan
- February 13, 2026: Received CE Mark approval (BSI NB 2797 under MDR 2017/745) for the InVera Infusion Device, enabling EU market entry
- March 12, 2026: Received FDA 510(k) clearance (K250794) for the InVera Infusion Device
- April 7, 2026: Public announcement of FDA clearance
- April 21, 2026: Scheduled presentation of 12-month pilot clinical study results by Dr. Lowell Kabnick at Charing Cross Vascular Symposium, London
Core Technology & Products
InVera Infusion Device (Flagship Product)
Device Description:
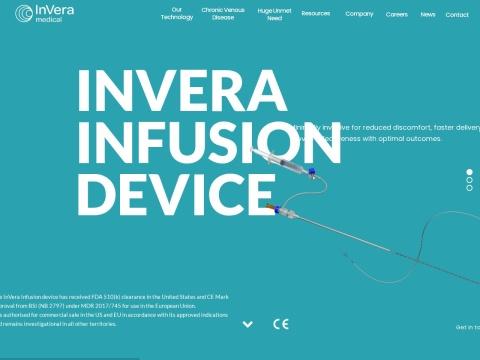
The InVera Infusion Device is a minimally invasive, single-use, disposable catheter system designed for the controlled infusion of physician-specified agents (including sclerosant) into the superficial veins of the lower limb. It represents the first device that mechanically prepares the vein wall to enhance sclerosant infusion to deeper layers.
Key Technical Specifications:
- Catheter Size: 5Fr (French gauge), 100cm working length
- Active Element: Nitinol helical coil (6mm diameter) with micro-textured lumen engaging surface composed of contiguous triangular features
- Deployment Mechanism: Pin-and-pull handle mechanism for deployment and recapture of the coil
- Imaging: Echogenic distal catheter section for ultrasound visualization during navigation
- Guidewire: Reinforced design allows direct access without guidewire requirement
- Sterilization: Single-use, disposable, sterile packaging
Mechanism of Action
The device works by:
1. Mechanical Abrasion: The helical coil's radial outward force and micro-textured surface stimulate venospasm, causing shrinkage of the target vein diameter
2. Wall Disruption: The abrasive element disrupts the inner media layer of the vein wall
3. Enhanced Infusion: Exposure of the subendothelial layer enables deeper penetration and more effective contact between sclerosant and vessel wall
4. Natural Closure: Results in natural scarring and vessel closure, diverting blood flow away from diseased vessels
Clinical Advantages
vs. Thermal Technologies (Radiofrequency/Laser):
- Eliminates need for multiple painful tumescent anesthesia injections
- Avoids risk of skin and nerve thermal injury
- Reduces bruising, swelling, and prolonged recovery periods
- Better suited for Office Based Lab (OBL) setting
vs. Current Non-Thermal Methods:
- Superior efficacy compared to standard sclerotherapy
- Non-implant alternative to cyanoacrylate glue embolization (avoids permanent foreign body implantation)
- Requires only single injection of local anesthesia vs. multiple injections
- Supports faster patient recovery
Regulatory Status & Approvals
FDA 510(k) Clearance (March 12, 2026)
- 510(k) Number: K250794
- Classification: Class II, Product Code KRA
- Regulation: 21 CFR 870.1210 (Continuous flush catheter)
- Predicate Device: ClariVein IC (K201907)
- Indications for Use: Infusion of physician-specified agents, including sclerosant, into veins of the peripheral vasculature (e.g., superficial veins, saphenous veins)
- Substantial Equivalence: Demonstrated through performance testing showing equivalent safety and effectiveness to predicate device
CE Mark Approval (February 13, 2026)
- Notified Body: BSI (British Standards Institution) NB 2797
- Regulation: MDR 2017/745 (Medical Device Regulation)
- Scope: European Union market authorization
Patent Portfolio
- US Patent 12,478,399: "Method of treating a blood vessel in a subject" (Granted November 25, 2025)
- Inventors: Sean Cummins (County Limerick), Stephen Cox (Westport), Nigel Phelan (County Galway)
- Technology: Covers the method of advancing catheter with deployable abrasive element, disrupting vessel wall, and exposing subendothelial layers
Clinical Evidence
Pilot Clinical Study
- Status: 12-month pilot study completed
- Presentation: Dr. Lowell Kabnick to present 12-month results at Charing Cross Vascular Symposium, London (April 21, 2026)
- Outcomes: Promising safety and procedural outcomes demonstrated
Market Opportunity
Disease Burden
- Prevalence: Chronic Venous Disease affects 1 in 4 adults (over 120 million people across the US and Europe)
- Treatment Gap: Only 1% of those living with venous disease currently receive interventional treatment annually
- Market Size: Over 1 million annual procedures in the US alone in the superficial venous reflux market (fastest growing non-thermal segment)
Corporate Information (2026)
Company Profile
Legal Name: InVera Medical Limited
Founded: July 24, 2018
Company Number: 630839 (Ireland)
Company Type: Private Limited Company
Status: Normal (Active)
Registered Address: Unit 113, Innovation Hub, Dublin Road, Galway H91 DCH9, Ireland
Principal Activity: Other Research and Experimental Development on Natural Sciences and Engineering
Company Size: Small Company
Shareholders: 16 shareholders
Website: www.inveramedical.com
Leadership Team
Stephen Cox: Chief Executive Officer and Co-Founder
Nigel Phelan: Chief Medical Officer and Co-Founder
Sean Cummins: Inventor and Key Technical Contributor
Innovation Ecosystem Affiliation
ATU iHubs: Client company of Atlantic Technological University innovation hubs
Achievement: Second ATU iHubs client company to receive FDA clearance
2026 Strategic Priorities & Outlook
Commercial Launch
- Limited Market Release: Initiating controlled launch in select US and European centers in 2026
- Market Segment: Targeting the fastest growing non-thermal segment of the superficial venous reflux market
- Clinical Adoption: Focusing on Office Based Lab (OBL) settings and outpatient vein clinics
Key Differentiators
- First-to-Market Mechanism: First device to mechanically prepare vein walls for enhanced sclerosant infusion
- Non-Thermal, Non-Implant: Unique position combining safety profile of non-thermal methods with efficacy approaching thermal ablation, without permanent implant risks
- Procedural Efficiency: Single anesthesia injection vs. multiple injections required by thermal methods
- Patent Protection: Strong IP position with granted US patent covering core mechanism of action
- Dual Regulatory Success: Simultaneous FDA and CE Mark approvals in 2026 enabling global commercial strategy
- Irish Innovation: Part of Ireland's growing medtech ecosystem with ATU iHubs support
Keywords: InVera Medical,chronic venous disease,CVD,venous infusion device,non-thermal catheter,sclerotherapy,varicose veins,Galway Ireland,Stephen Cox,Nigel Phelan,FDA 510(k),CE Mark,helical coil,K250794,ClariVein,medical device,ATU iHubs,Dr. Lowell Kabnick
猜你喜欢
-
Elumn8 Medical - Image-Guided Coronary Intervention Pioneer
Elumn8 Medical(formerly Simpson Interventions) is a me... -
Shanghai Sanyou Medical Co., Ltd. (上海三友医疗器械股份有限公司) - Leading Chinese Orthopedic Implant Manufacturer
Shanghai Sanyou Medical Co., Ltd. (stock code: 688085)... -
Robocath S.A.S (罗博凯思公司) - French Pioneer in Vascular Intervention Robotics
Robocath S.A.S (罗博凯思公司) is a French medical robotics company... -
Stereotaxis, Inc. (美国思瑞泰心国际公司) - Global Leader in Robotic Magnetic Navigation for Cardiac Interventi
Stereotaxis, Inc. (NYSE: STXS), known in Chinese markets as... -
Sonire Therapeutics Inc. - Pioneering Next-Generation HIFU Therapy for Pancreatic Cancer
Sonire Therapeutics Inc. is a clinical-stage medical d... -
InVera Medical - Irish Medical Device Innovator in Chronic Venous Disease Treatment
InVera Medical is an Irish medical technology company founde... -
1st SurgiConcept (第一外科概念) - French Aesthetic Thread and Medical Device Specialist
1st SurgiConcept (第一外科概念公司) is a French medical device manuf... -
10x Genomics, Inc. - Pioneer in Single Cell and Spatial Biology Technologies
10x Genomics, Inc. (Nasdaq: TXG) is a life science technolog... -
Crystal Optic Co., Ltd. (주식회사 크리스탈옵틱) - Korea's Premier Custom Surgical Loupe Manufacturer
Crystal Optic Co., Ltd. (주식회사 크리스탈옵틱) is a South Korea... -
Warantec Co., Ltd. (주식회사 워랜텍) - Korea's Leading Dental Implant Manufacturer with Academic Heritage
Warantec Co., Ltd. (워랜텍) is a South Korean dental implant ma... -
Endovision Co., Ltd. (엔도비전) - Korea's Chitosan-Based Biomaterial Platform Pioneer
Endovision Co., Ltd. (엔도비전) is a South Korean medical devic... -
Outlook Surgical
Outlook Surgical, based in Vancouver, Canada, develops advan...
网友评论
- 热门标签
-
- USA
- California
- China
- Germany
- Korea
- France
- Shanghai
- Florida
- New York
- Zhejiang
- Robocath
- Zimmer
- Indiana
- Medtronic
- Mentor
- Johnson
- Munich
- Massachusetts
- Canada
- British Columbia
- Ningbo
- Ireland
- Seoul
- Michigan
- Yokneam
- Elumn8
- Israel
- Pulse
- PillSafe
- Oklahoma
- SBE
- Ronovo
- Diality
- Dermalogica
- Womed
- Occitanie
- INT Medical
- enVVeno
- Medline
- Northfield
- Illinois
- Brainlab
- RISTEK INSTRUMENTS
- Sialkot
- Pakistan
- Siemens
- Sigma-Laborzentrifugen
- Harz
- InVera
- Hauts-de-France
- 关注我们
-

扫一扫二维码关注我们的微信公众号


