In October 2023, Advanced Bifurcation Systems received the U.S. FDA Breakthrough Device Designation for its bifurcation stenting technology. In January 2025, the company closed an oversubscribed USD 20.8 million SAFE financing round and expanded its Board of Directors with Malia Chui and SUSAn Yashar. On April 27, 2026, ABS acquired all assets of Svelte Medical Systems, Inc., including drug-eluting coronary stents, all intellectual property, and comprehensive U.S. and international regulatory approvals (FDA PMA, CE Mark, Japan PMDA). This acquisition places ABS in a small group of select global companies possessing an FDA-approved drug-eluting coronary stent. The company has submitted a proposal to the FDA for a clinical study leADINg to an IDE for its bifurcation technology, with FDA approval targeted for late 2026.
Company Profile (Updated April 2026)
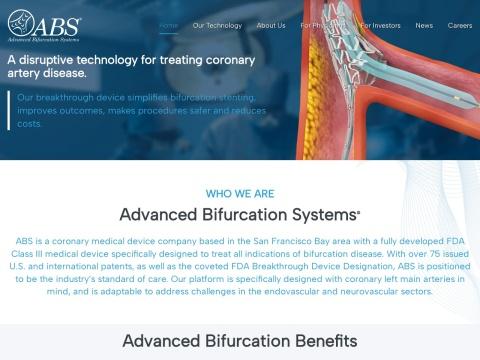
Advanced Bifurcation Systems Inc. (ABS) is a privately held medical device company headquartered in Livermore, California, USA (San Francisco Bay Area). Founded in 2010 by Dr. Mehran J. Khorsandi (cardiologist, Cedars Sinai Medical Center) and Henry Bourang (renowned medical devices engineer), ABS is dedicated to addressing the challenges of coronary bifurcation disease through an innovative, minimally invasive stent delivery platform. The company has developed a Class III medical device specifically designed to treat all indications of bifurcation disease, with coronary left main arteries as a Primary focus, and is adaptAble to address challenges in the endovascular and neurovascular sectors. The company currently employs 12 people.
Development History:
- 2010: Founded in Livermore, CA, by Dr. Mehran Khorsandi and Henry Bourang
- 2018: Completed accelerator/incubator program financing
- December 2020: Closed USD 11 million Series A financing round
- 2021-2022: Completed first-in-human trials across three international medical centers with 100% success rate
- September 2023: Completed later-stage venture financing
- October 2023: Received U.S. FDA Breakthrough Device Designation
- January 2025: Closed USD 20.8 million SAFE financing (oversubscribed); expanded Board of Directors
- 2025: Submitted proposal to U.S. FDA for clinical study leading to IDE approval
- April 2026: Acquired all assets of Svelte Medical Systems, Inc., including FDA-approved drug-eluting coronary stents and international regulatory approvals
Core Mission: To transform the treatment of coronary bifurcation lesions by providing an innovative solution that simplifies the procedure, improves patient outcomes, reduces the need for repeat interventions and open-heart surgery, and enables all interventionalists—not just experts—to confidently perform bifurcation stenting.
2026 Breakthrough: Acquisition of Svelte Medical Systems
Strategic Acquisition (April 27, 2026)
Transaction Overview:
- On April 27, 2026, ABS announced the acquisition of all assets of Svelte Medical Systems, Inc.
- Acquired Portfolio: Drug-eluting coronary stents, all intellectual property, and related comprehensive U.S. and international regulatory approvals and filings
- Regulatory Assets: U.S. FDA Premarket Approval (PMA); Europe CE Mark; Japan PMDA approval
- Strategic Significance: Places ABS in a small group of select global companies possessing an FDA-approved drug-eluting coronary stent
- Synergy: Combined with ABS's innovative bifurcation platform (FDA Breakthrough Device Designation), the acquisition accelerates ABS's development pathway and materially strengthens its presence in the global interventional cardiology market
- CEO Statement: "As we advance our mission to transform the treatment of coronary bifurcation lesions, this acquisition will expand our commercial portfolio and represents a pivotal strategic milestone," said Dr. Mehran Khorsandi, Chairman and CEO of ABS
Core Product: ABS Bifurcation Stent Delivery Platform
ABS System Technology
- Design Philosophy: Unlike past attempts that focused solely on the stent, the ABS System focuses on the delivery platform, providing a groundbreaking proprietary stent delivery system
- Platform: Modular, independently movable dual-catheter system (mother-daughter configuration) for provisional side-branch stenting
- Material: Stainless steel platform with 105-micron strut thickness
- Configurations:
- ABS MD-P (Provisional): Single stent on the mother catheter; daughter catheter has no stent mounted, used for provisional side-branch stenting
- ABS MD-Bi (Bifurcating): Mother-daughter stent on the mother catheter plus a daughter stent on the daughter catheter for upfront two-stent strategy
- Stent Sizes: Both MDS and DS available in sizes ranging from 2.5mm to 4.0mm; every combination between these two is possible
- Stent Length: MDS 18mm, DS 8mm
- Guiding Catheter: 6 Fr compatible, 7 Fr preferred
- Key Innovation: Enables provisional stenting without rewiring or stent deformation; simultaneous side branch and proximal main branch stent deployment ensures perfect alignment without gaps or overlaps at the side branch ostium
- Deployment: System loaded on two wires, advanced to the carina, assembled using the fixed structure of the carina for automatic alignment, followed by sequential balloon inflation and kissing inflation
- Coating: Currently available in bare metal only; a sirolimus-coated version with biodegradable polymer is under development
Clinical Evidence
- First-in-Human (FIM) Study: Pilot, non-randomized, multicenter trial across three international medical centers enrolling 10 patients
- Procedural Success: 100% delivery rate and procedural success; no residual stenosis by IVUS; good stent apposition with complete coverage of the carina
- Safety: 30-day composite MACE (death, MI, target vessel revascularization): 0%
- Durability: 12-month composite MACE and TVR reMained at 0%; all patients free of angina
- Follow-Up: Angiographic follow-up at 6 months completed in 7 patients; revealed edge restenosis in two side branch vessels only
- Lesion Types: MD-Bi deployed in LAD-diagonal, LCx-obtuse marginal, and PDA-PLB bifurcations; MD-P deployed in LAD-diagonal; all lesions were Medina 1,1,1 by visual estimation
- Ongoing Development: Human studies including left main (LM) bifurcations are in the planning stages
Financial and Funding History
Funding Rounds
- Accelerator/Incubator (Jun 2018): Initial program completion
- Series A (Dec 2020): USD 11 million raised
- Later Stage VC (Sep 2023): Clinical trials stage financing completed
- SAFE Financing (Jan 2025): USD 20.8 million oversubscribed round from high-net-worth individuals and family offices across the U.S. and internationally
- Total Raised: Approximately USD 40 million to date
- Investors: Brandon Holdings, LLC (largest investor through SAFE), Capital Factory, Cedars-Sinai, MaRS Investment Accelerator Fund
Company Leadership and Governance
Executive Team
Dr. Mehran J. Khorsandi - Chairman & Chief Executive Officer:
- Cardiologist at Cedars Sinai Medical Center
- Co-founder of ABS
Henry Bourang - President & Chief Operating Officer:
- Renowned medical devices engineer
- Co-founder of ABS
Ken Richards - Chief Administrative Officer / Chief Financial Officer:
- Oversees administrative and financial operations
Board of Directors
- Malia Chui: Represents Brandon Holdings, LLC (ABS's largest investor); brings decades of expertise in investment management with specialization in healthcare, including hospital ownership and operations
- Susan Yashar: Recently retired as Deputy Global CEO and General Counsel of Deloitte Global; previously served in senior roles at the Securities & Exchange Commission and the Public Company Accounting Board; brings deep experience in regulatory compliance, corporate governance, and international business
Scientific Advisory Board
- Willie Lawrence, MD: New member of Scientific Advisory Board and Advisor to the CEO; instrumental in guiding FDA and international regulatory processes
Global Market Position and Competition
Market Landscape
Coronary Bifurcation Disease Burden:
- Coronary bifurcation lesions occur in approximately 20% of all Percutaneous Coronary Intervention (PCI) procedures and 20% of Coronary Artery Bypass Grafting (CABG) procedures
- Represents a significant addressable market in interventional cardiology
- Patients currently face limited options: complex stenting procedures with suboptimal outcomes, or open-heart CABG surgery with higher risks and costs
Primary Competitors
- Vesper Medical: Competitor in bifurcation stenting
- Poseidon Medical: Competitor in bifurcation stenting
- Medtronic: RESOLUTE ONYX Post-Approval Study includes bifurcation cohort; FDA-approved for non-left main bifurcation lesions using provisional stenting
- Tryton Medical: Tryton Side Branch Stent for coronary bifurcations
- Cordis (Cardinal Health): Comprehensive interventional cardiology portfolio
Competitive Advantages
- Delivery Platform Focus: Unlike competitors focusing only on stent design, ABS innovates the entire delivery platform for reproducible, predictable outcomes
- Provisional Stenting Without Rewiring: Unique ability to perform provisional side-branch stenting without rewiring or stent deformation
- Carina-Based Alignment: Uses the only fixed (non-variable) structure in any bifurcation—the carina—for automatic, perfect stent alignment
- Simultaneous Deployment: Only system with simultaneous side branch and proximal main branch stent deployment ensuring full tissue coverage without gaps or overlaps
- Hybrid Monorail/OTW System: Minimizes wire-wrap and permits easier handling
- Physician-Founded: Developed by a practicing cardiologist and veteran engineer with deep clinical and technical insight
- Dual Portfolio: Through the Svelte acquisition, ABS now possesses both a breakthrough bifurcation platform and an FDA-approved drug-eluting stent
- Platform Expandability: Adaptable to endovascular and neurovascular applications beyond coronary bifurcation
Regulatory Status and Clinical Pathway
FDA and International Regulatory Progress
- Breakthrough Device Designation: Received from U.S. FDA in October 2023
- IDE Submission: Proposal submitted to FDA for clinical study leading to IDE approval
- FDA Approval Target: Late 2026
- Device Classification: FDA Class III medical device
- Commercial Availability: Device is not yet available for commercial use; all safety and efficacy claims are based on ongoing research and have not been validated by regulatory authorities
- Svelte Legacy Approvals: Through the April 2026 acquisition, ABS now holds FDA PMA, CE Mark, and Japan PMDA approvals for drug-eluting coronary stents
2026 Outlook and Strategic Priorities
Clinical and Regulatory Roadmap
- IDE Approval: Pursue FDA IDE approval for bifurcation technology clinical study
- Pivotal Trial: Conduct U.S. pivotal clinical trial to support PMA submission
- FDA Approval: Target late 2026 for ABS bifurcation device FDA approval
- Left Main Expansion: Advance planning for human studies in left main (LM) bifurcations
- Drug-Eluting Version: Continue development of sirolimus-coated system with biodegradable polymer
Commercial and Strategic Expansion
- Svelte Integration: Leverage acquired FDA-approved drug-eluting stent portfolio to accelerate commercial readiness
- Global Market Entry: Prepare for commercial launch in the U.S. and expansion to European and Japanese markets leveraging Svelte's regulatory footprint
- Platform Diversification: Explore applications in endovascular and neurovascular sectors
- Series B Financing: Continue fundraising activities to support clinical development and commercialization
Contact Information
Headquarters
Address: 7419 Southfront Road, Livermore, California 94551, USA
Phone: 877-413-5845
Website: www.advancedbifurcation.com
Investor Relations
Status: Private company
Email: info@advancedbifurcation.com
Corporate Website: www.advancedbifurcation.com
Keywords: Advanced Bifurcation Systems, ABS, bifurcation stent, coronary bifurcation, PCI, CABG, interventional cardiology, stent delivery platform, Mehran Khorsandi, Henry Bourang, Livermore, California, FDA Breakthrough Device, IDE, drug-eluting stent, Svelte Medical, Class III, medical device, coronary artery disease, mother-daughter catheter, provisional stenting, carina alignment, sirolimus, bare metal stent, left main, Brandon Holdings, Malia Chui, Susan Yashar, Willie Lawrence