
In 2025, AIOBIO reported revenue of KRW 3.43 billion (+46.16% YoY). In March 2026, the company secured KRW 1 billion in funding from ANI CO., LTD. The company held its Annual General Meeting on March 31, 2026. AIOBIO's QLF™ technology-based devices are covered by Korean national health insurance for children aged 5-12. The Qraycam PRO received FDA 510(k) clearance (K221275) in September 2024 as a Class II Laser Fluorescence Caries Detection device. The company's products are now used in 800+ hospitals and clinics worldwide.
Company Profile (Updated April 2026)
AIOBIO Co., Ltd. (아이오바이오) is a South Korean healthcare technology company dedicated to revolutionizing Dental care through early diagnosis and preventative care. Founded in August 2011 and headquartered in Seoul, South Korea, the company is listed on the Korea KONEX market (Stock Code: 447690).
AIOBIO is a pioneer in Quantitative Light-induced Fluorescence (QLF™) technology, which enAbles early detection of dental caries, plaque, tartar, fissures, and other oral health issues that are difficult to identify with the naked eye or traditional X-rays. The company's vision is to become a total dental service and solutions provider embracing disease prevention, diagnosis, treatment, and post-treatment maintenance.
Core Mission: "At AIOBIO, we set new standards in oral health by introducing innovative diagnostic and examination methods, ultimately striving to enhance overall well-being and happiness. Our technology goes beyond traditional oral checkups, offering precise and effective diagnostic solutions that earn the trust of both patients and dental professionals."
Founder and Leadership
Co-Founder, CEO & Chairman: Dr. Hong-Cheol Yoon (윤홍철)
- Dentist with a doctorate in dental medicine from Yonsei University, Korea
- Opened his own dental clinic in 1998 during the Asian Financial Crisis
- Met Dutch physicist Dr. Elbert de Josselin de Jong in 2005 and co-founded AIOBIO in 2011
- Vision: "Just as you take X-rays when you have issues with your bones or muscles, people will be seeking diagnoses through our devices when they have issues with their teeth."
Co-Founder & Technical Advisor: Dr. Elbert de Josselin de Jong
- Dutch physicist and inventor of QLF™ (Quantitative Light-induced Fluorescence) technology
- Discovered during his PhD research at the University of Groningen in the 1980s that light could be used to identify dental decay
- First introduced the QLF™ prototype at a dentistry conference in Germany in 1993
- Moved to Korea in 2007 to collaborate on commercializing QLF™ technology
Core Technology
QLF™ (Quantitative Light-induced Fluorescence)
QLF™ technology uses 405-nanometer visible blue light (as opposed to radioactive rays used in X-rays) to examine dental plaque, tartar, decay, fissures, and other dental problems. When blue light is shone on teeth, healthy enamel and damaged enamel reflect the light at different wavelengths. Once the reflected light is examined using special film, only the damaged part displays bright red fluorescence.
Key Advantages:
- Early Detection: Identifies dental symptoms at very early stages before they become visible to the naked eye or detectable by X-ray
- Radiation-Free: Uses visible blue light instead of radioactive rays, making it safe for all patients including children and pregnant women
- Quantitative Analysis: Software quantifies oral hygiene status using four variables: ΔF, ΔF max, ΔR, ΔR max
- Patient Education: Visual fluorescence images make it easier for patients to understand their oral health condition
QBLISS™ (Biofluorescence Light Imaging and Screening Solution)
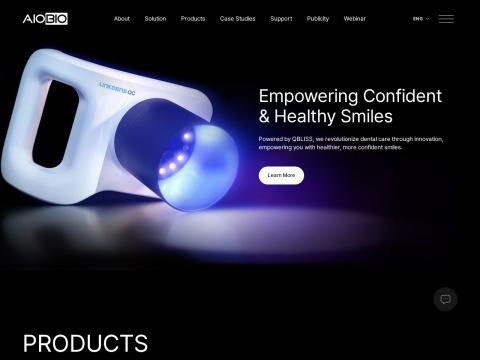
QBLISS™ harnesses advanced biofluorescence imaging to deliver augmented reality-style visualization free of chemical disclosing agents. It enables early detection and precise evaluation of oral disease, enhancing diagnostic accuracy and empowering clinicians to act with confidence.
Product Portfolio
Professional Clinical Devices
- Qraycam Pro (LINKDENS QC): A chairside diagnostic camera designed for quick biofilm detection and oral hygiene assessment. Ideal for routine checkups and patient education. FDA 510(k) cleared (K221275, September 2024) as a Class II Laser Fluorescence Caries Detection device (21 CFR 872.1745)
- Qraypen C: An intraoral imaging camera that captures high-resolution visuals for documentation, diagnosis, and patient communication. Features white, blue, and Orange light modes. FDA 510(k) cleared (K183121, July 2019) as a Class I Dental Intraoral Camera (21 CFR 872.6640) for oral cavity video imaging
- LINKDENS VU: A compact, handheld device that uses fluorescence to visualize plaque and calculus in real time, perfect for hygiene visits and patient motivation
- Qrayview C: Medical device Class 2 approved in Korea (December 2017)
Home Use and Portable Devices
- Qscan Plus: A portable diagnostic scanner designed for home use, ideal for caregivers, parents, and older adults. Allows for oral health monitoring between dental visits
- Qraypen W-Block: Home-use version of the QLF diagnostic device
Software and AI Platform
- Qray SW (Qray Software): Analyzes images from Q-ray devices using four quantitative variables (ΔF, ΔF max, ΔR, ΔR max) to establish precise diagnosis and treatment plans. Features Simple Hygiene Scores (SHS) analysis for plaque distribution and brushing habit training
- LINK DENS Platform: An AI-driven digital platform connecting clinics and patients through real-time data sharing, instant imaging analysis, and remote patient monitoring
Certifications and Regulatory Milestones
- FDA 510(k) Clearance — Qraycam PRO: Received FDA 510(k) clearance in September 2024 (K221275) as a Class II Laser Fluorescence Caries Detection device (21 CFR 872.1745) for early caries detection
- FDA 510(k) Clearance — Qraypen: Received FDA 510(k) clearance in July 2019 (K183121) as a Class I Dental Intraoral Camera (21 CFR 872.6640) for oral cavity video imaging
- Korean Medical Device Class 2 Approval: Qrayview C (December 2017), Qraypen C and Qray Software (January 2018)
- New Health Technology Assessment: Passed Korea's Ministry of Health and Welfare New Health Technology Assessment in August 2018 — the first-ever approval for such a dental diagnostic device under the new assessment system
- National Health Insurance Coverage: From June 2021, patients aged 5 to 12 can receive public health insurance benefits every six months for dental check-ups using QLF™ devices (patients pay approximately KRW 4,000 / ~$3.59 per visit)
- Venture Business Certification: Obtained in 2013
- Promising Startup: Selected by the Ministry of SME and Startups in 2018
- Citation from Minister of Health and Welfare: Received in 2016 for contributing to the development of health and medical technology
2025-2026 Latest Developments
Financial Performance (2025)
Revenue (2025): KRW 3.43 billion (approximately $2.4 million), up 46.16% year-over-year
Net Income (2025): Loss of approximately KRW 5.3 billion
Revenue Growth: Significant acceleration from previous years, driven by expanded product adoption and international sales
Stock Price (April 2026): Approximately KRW 3,250
Market: Korea KONEX (447690)
Funding and Corporate Events (2026)
- March 16, 2026: Announced receipt of KRW 1 billion in funding from ANI CO., LTD
- March 31, 2026: Held Annual General Meeting
- January 10, 2025: Funding transaction from ANI CO., LTD approved by shareholders with a 3% maturity rate
Strategic Expansion
- Global Distribution: Products exported to Australia, India, Mexico, Russia, and The Netherlands, with plans for further expansion
- US Market: Qraycam PRO and Qraypen series FDA-cleared; distribution through partners such as KB Dental Products Inc. (California)
- 5 Step Dentistry System: Launched in 2018 after passing the New Health Technology Assessment, covering screening, diagnosis, assessment, treatment, and post-treatment maintenance
- AI and Big Data Platform: Developing a digital platform for personal dental healthcare using AI and big data, with government funding from the Ministry of Health and Welfare until 2023
Company Basic Information
Corporate Overview
Company Full Name: AIOBIO Co., Ltd. (아이오바이오)
Founded: August 2011
Co-Founders: Dr. Hong-Cheol Yoon (CEO) and Dr. Elbert de Josselin de Jong (Technical Advisor)
Headquarters: 8F, 38, Teheran-ro 4-gil, Gangnam-gu, Seoul, 06232, Republic of Korea
Factory: #306, Sunil Technopia, 555 Dunchon-daero, Jungwon-gu, Seongnam-si, Gyeonggi-do, 13215, Republic of Korea
Stock Exchange: Korea KONEX
Stock Code: 447690
Company Type: Public Listed Company (KONEX)
Industry: Medical Equipment / Dental Diagnostics / Healthcare Technology
Employees: 44 (as of 2025)
Official Website: www.aiobio.com / www.aiobio.co.kr
Shop: shop.aiobio.com
Email: aiobio@aiobio.co.kr / support@aiobio.com
Telephone: +82-31-698-4991 / +1 (917) 275-7862 (US support)
Production Facilities
- Wonju Factory: Built in 2013 at Wonju Medical Industry Techno Valley
- Seongnam Factory: Built in 2016 at Sunil Technopia, Seongnam
Key Investors and Funding History
- 2016: Investment contract with Ever Green Investment Partners
- 2016: Investment from DSC Investment and Ever Green Investment Partners
- 2019: Issued KRW 4.5 billion in redeemable convertible preference shares (RCPS), purchased by DSC Investment, Korea Investment Partners, and IMM Investment
- 2026: KRW 1 billion funding from ANI CO., LTD
- Individual Investors: Approximately 30 medical professional investors providing technical advice and business partnerships
Market Presence and Competitive Landscape
Domestic Market (South Korea)
- Target Market: 18,000 dental hospitals and clinics in Korea; AIOBIO targets supplying to 3,600 (20%) of them
- Current Coverage: 800+ hospitals and clinics globally using QLF™ devices
- Insurance Coverage: National health insurance covers QLF™ check-ups for children aged 5-12 every six months
International Market
- Export Markets: Australia, India, Mexico, Russia, The Netherlands, United States, Japan, and other countries
- US Distribution: Partnership with KB Dental Products Inc. (Placentia, California)
- Europe: Export contracts with Inspektor in the Netherlands
- Asia: Export contracts with ITO Group in Japan, CI Medical in Japan, and Russia Vysota
Competitive Position
AIOBIO is recognized as a leADINg global pioneer in biofluorescence dental diagnostics. The company creates a new market segment in dental diagnosis, which traditionally accounts for only about 10% of total dental payments in Korea (compared to 70% in the US). By driving growth in the diagnosis sector, AIOBIO aims to transform dental care from treatment-focused to prevention-focused.
2026 Outlook and Strategic Priorities
Strategic Focus Areas
- Technology Prevalence: Make QLF™ technology the industry norm for dental diagnosis, both in Korea and internationally
- AI Platform Development: Build a digital platform for personal dental healthcare using AI and big data, enabling patients to self-manage dental data
- Home Healthcare Expansion: Expand QLF™ devices from clinical settings to household use, targeting 2,000 household customers
- Global Partnerships: Collaborate with global oral care product companies to create comprehensive dental services combining diagnostic equipment with traditional oral care products
- B2B Sales Growth: Expand sales to dental clinics and hospitals by raising brand and technology awareness
- Regulatory Expansion: Pursue additional product approvals from US and European authorities
Competitive Advantages
- Original Technology: Global patent-protected QLF™ platform technology invented by Dr. Elbert de Josselin de Jong and commercialized by AIOBIO
- First-Mover Advantage: First company to pass Korea's New Health Technology Assessment for dental diagnostic devices
- Radiation-Free Safety: Visible blue light technology safe for all patients, including children
- Insurance Coverage: Only dental diagnostic technology covered by Korean national health insurance for pediatric patients
- Dual B2B/B2C Model: Revenue from both professional clinical devices and home-use consumer products
- AI Integration: LINK DENS platform and Qray SW software provide AI-driven diagnostics and remote patient monitoring
Risk Factors
- Market Education: Need to change cultural perception that dental visits are only necessary when in pain
- Regulatory Hurdles: New Health Technology Assessment and international regulatory approvals require significant clinical data and time
- Competition: Traditional X-ray and visual inspection methods dominate dental diagnosis
- Financial Performance: Company reported net losses; need to ACHieve sustainable profitability
- Macroeconomic: COVID-19 and global economic conditions may impact overseas sales expansion
Contact Information
Headquarters
Address: 8F, 38, Teheran-ro 4-gil, Gangnam-gu, Seoul, 06232, Republic of Korea
Tel: +82-31-698-4991
Email: aiobio@aiobio.co.kr
Website: www.aiobio.com
Factory
Address: #306, Sunil Technopia, 555 Dunchon-daero, Jungwon-gu, Seongnam-si, Gyeonggi-do, 13215, Republic of Korea
US Support
Tel: +1 (917) 275-7862
Email: support@aiobio.com
US Distribution Partner
KB Dental Products Inc.
177 W. Orangethorpe Ave, Placentia, CA 92870, USA
Email: kbdental@att.net
Keywords: AIOBIO,아이오바이오,447690,KONEX,Dental Diagnostics,QLF,Quantitative Light-induced Fluorescence,Q-ray,Qraycam Pro,Qraypen C,Qscan Plus,LINKDENS VU,QBLISS,Biofluorescence,Oral Health,Early Caries Detection,Hong-Cheol Yoon,Elbert de Josselin de Jong,Seoul,South Korea,FDA 510(k),K221275,K183121,New Health Technology Assessment,National Health Insurance
You May Also Like
-
AIOBIO Co., Ltd. - AI-Powered Dental Diagnostics Pioneer
AIOBIO Co., Ltd. is a South Korean healthcare technology com... -
Tandem Diabetes Care, Inc. - Leading Insulin Delivery and Diabetes Technology Innovator
Tandem Diabetes Care, Inc. is a global insulin delivery and... -
Beijing Coyote Bioscience Co., Ltd. (北京卡尤迪生物科技股份有限公司) - Pioneer in POCT Molecular Diagnostics
Beijing Coyote Bioscience Co., Ltd. (北京卡尤迪生物科技股份有限公司) is a n... -
Danaher Corporation (丹纳赫公司) - Global Life Sciences and Diagnostics Innovator
Danaher Corporation (丹纳赫公司) is a leading global life science... -
Zybio Inc. (中元汇吉生物技术股份有限公司) - Leading Chinese Full-Line IVD Solutions Provider
Zybio Inc. (中元汇吉生物技术股份有限公司) is one of China's leading in vit... -
Guangzhou Weimi Bio-Tech Co., Ltd. (广州市微米生物科技有限公司) - Innovative Chinese IVD and POCT Solutions Provi
Guangzhou Weimi Bio-Tech Co., Ltd. (广州市微米生物科技有限公司) is a nati... -
Zhuhai Keyu Biological Engineering Co., Ltd. (珠海科域生物工程股份有限公司) - Leading Chinese IVD Automation Solut
Zhuhai Keyu Biological Engineering Co., Ltd. (珠海科域生物工程股份有限公司... -
Autobio Diagnostics Co., Ltd. (郑州安图生物工程股份有限公司) - Leading Chinese IVD Solutions Provider
Autobio Diagnostics Co., Ltd. (郑州安图生物工程股份有限公司) is one of Chi... -
Aiforia Technologies Plc - Leading AI-Powered Digital Pathology Solutions Provider
Aiforia Technologies Plc is a publicly traded Finnish medica... -
Dermalogica - Global Professional Skincare Leader
Dermalogica is the world's leading professional skincare bra... -
DermaSensor Inc. - AI-Powered Handheld Skin Cancer Detection Pioneer
DermaSensor Inc. is a Miami-based medical technology company... -
Wuhan Easy Diagnosis Biomedicine Co., Ltd. (武汉明德生物科技股份有限公司) - Leading Chinese IVD and POCT Solutions
Wuhan Easy Diagnosis Biomedicine Co., Ltd. (武汉明德生物科技股份...
网友评论
- Popular Tags
-
- USA
- California
- China
- Germany
- Korea
- Abbott
- Massachusetts
- Shanghai
- Florida
- Israel
- Guangdong
- Japan
- Zhejiang
- Illinois
- QIAGEN
- ARKRAY
- France
- Ireland
- Beijing
- British Columbia
- Canada
- Seoul
- Michigan
- Jiangsu
- Arizona
- Netherlands
- 3M
- Minnesota
- North Rhine-Westphalia
- Singapore
- Ningbo
- Washington
- New York
- 爱科来
- Medtronic
- Finland
- Munich
- Suzhou
- Agilent
- Tokyo
- Mentor
- Missouri
- Johnson
- SynCardia
- Fujirebio
- Gyeonggi-do
- Robocath
- Autobio
- 安图生物
- Henan
- Follow Us
-

Scan the QR code to follow our WeChat Official Account

